 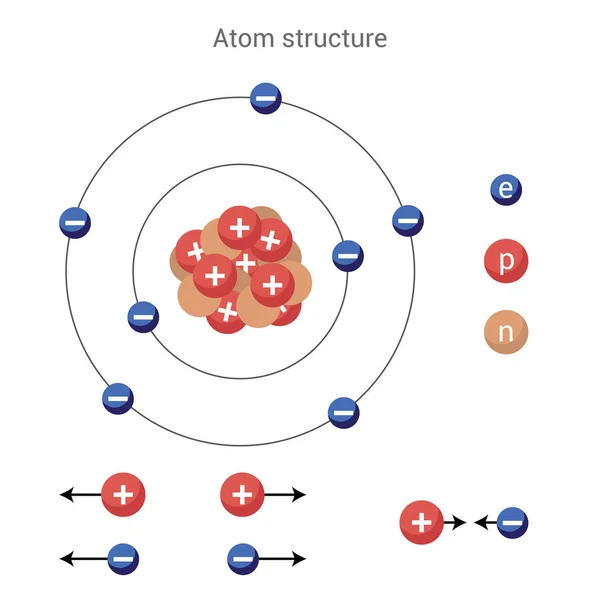
Ε 0 ℏ c īy combining the best measured value of the antiproton charge (below) with the low limit placed on antihydrogen's net charge by the ALPHA Collaboration at CERN. See also: 2019 redefinition of the SI base units Elementary charge Detailed measurements have shown that the magnitude of the charge of the proton. Under assumptions of the then-disputed atomic theory, the elementary charge had also been indirectly inferred to ~3% accuracy from blackbody spectra by Max Planck in 1901 and (through the Faraday constant) at order-of-magnitude accuracy by Johann Loschmidt's measurement of the Avogadro number in 1865. The electric charge of electrons, protons and neutrons are listed in Table 22.2. 2 This elementary charge is a fundamental physical constant. Millikan and Harvey Fletcher's oil drop experiment first directly measured the magnitude of the elementary charge in 1909, differing from the modern accepted value by just 0.6%. The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge 1 e. 
In the centimetre–gram–second system of units (CGS), the corresponding quantity is 4.803 2047. Since the 2019 redefinition of SI base units, the seven SI base units are defined by seven fundamental physical constants, of which the elementary charge is one. The two particles exert forces on each other, with the force decreasing as the distance between them. atoms See all videos for this article More than 90 types of atoms exist in nature, and each kind of atom forms a different chemical element. In the SI system of units, the value of the elementary charge is exactly defined as e e = 1.602 176 634 ×10 −19 coulombs, or 160.2176634 zeptocoulombs (zC). The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric. Step 1: Calculation for electron of charge (part a). The proton’s positive charge is equal and opposite to the negative charge on an electron, meaning a neutral atom has an equal number of protons and electrons. The attraction between the negative charge of an electron, and the postive charge of the proton, is what keeps the electrons in orbit around the nucleus. This elementary charge is a fundamental physical constant. The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge −1 e. The proton’s positive charge is equal and opposite to the negative charge on an electron, meaning a neutral atom has an equal number of protons and electrons. Charge carried by one proton or electron Elementary charge 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
